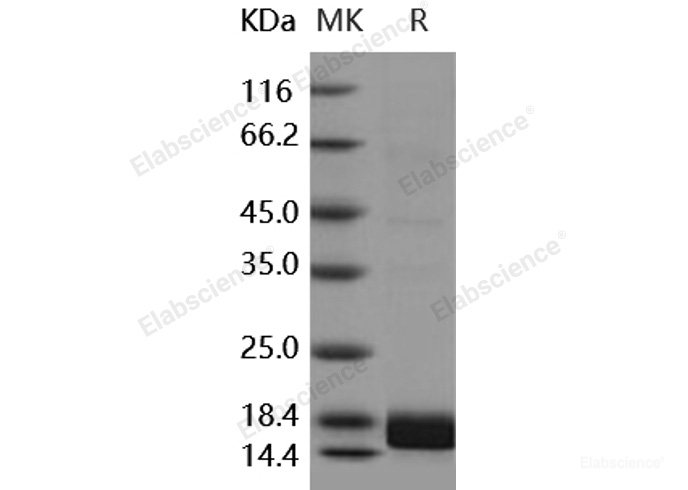
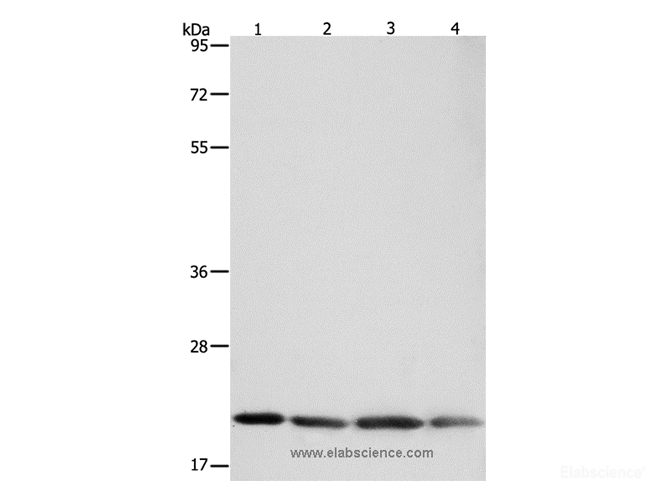
- HOME
-
PRODUCTS
- Hot
- QuicKey Pro ELISA
- High Sensitivity ELISA Kits
- Monkeypox Test Kit
- Uncoated ELISA Kits
- Fluorometric Assay Kit
- TOS and TAS Assay Kits
- IHC Validated Antibodies
- Cancer Research Antibodies
- TUNEL Assay Kit
- Immunology Related Reagents
- Antibodies
- Flow Cytometry Antibodies
- Polyclonal Antibody
- Monoclonal Antibody
- Virus Antibody
- Secondary Antibody
- KO Validated Antibodies
- Acetyl Phospho Methyl Antibody
- Ready-to-Use Antibodies
- Rabbit Monoclonal Antibody
- SERVICES
-
Cell Types
- T/B/NK Cell Population Detection
- Detection of T/B/NK (4-color) in human peripheral blood
- Detection of T/B/NK (4-color) in C57BL/6 mouse peripheral blood
- Detection of TB cells (4-color) in mouse lymph node
- Detection of T/B/NK (5-color) in Rat Spleen
- Detection of T/B/NK (3-color) in Rat Spleen
- CD4+/CD8+T Cell Population Detection
- Detection of T cells (4-color) in Human Peripheral Blood
- Detection of T Cells (2-color) in Mouse Thymus
- Detection of Lymphocyte (4 colors) in Mouse Tumor
-
Research
- Research Areas
- Cancer
- Cardiovascular
- Cell Biology
- Developmental Biology
- Epigenetics and Nuclear Signaling
- Immunology
- Metabolism
- Microbiology
- Neuroscience
- Signaling transduction
- Stem Cells
- Tags and Cell Markers
- Pathways
- Akt/PKB Signaling Pathway
- AMPK Signaling Pathway
- EGFR Signaling Pathway
- JAK/STAT Signaling Pathway
- MAPK-ERK Signaling Pathway
- MAPK-JNK Signaling Pathway
- MAPK-p38 Signaling Pathway
- Inhibition of MMPs
- mTOR Signaling Pathway
- NF-kB Signaling Pathway
- P53 Signaling Pathway
- Regulation of Apoptosis
- TGF-β Signaling Pathway
- VEGF Signaling Pathway
- Wnt Signaling Pathway
- RESOURCES
- ABOUT US
- SARS-CoV-2